All published articles of this journal are available on ScienceDirect.
Solubility of Bioactive Substances in Ethyl Lactate + Water Mixtures: Ferulic Acid and Caffeine
Abstract
The solubility of ferulic acid in ethyl lactate + water mixtures was measured at ambient temperature and pressure, and reported for the first time in this work. It was demonstrated that the acid solubility in the respective mixed solvents was considerably higher than in either pure ethyl lactate or water. Similar behavior pattern was recently reported by the authors for the solubility of caffeine in ethyl lactate + water mixtures.
UNIQUAC and modified UNIFAC (Dortmund) were applied to represent the solubility of ferulic acid and caffeine in the mixed solvents and their capabilities to describe adequately the cosolvent effect observed was tested and compared. The results obtained demonstrated that the UNIQUAC model provided excellent correlation of the experimental data for both systems. The UNIFAC model, on the other hand, was applied in a completely predictive manner and produced very good results for ferulic acid solubility and just qualitative predictions for caffeine.
1. INTRODUCTION
Ferulic acid and caffeine Fig. (1) are bioactive compounds derived from natural sources which draw commercial attention to the development of high value-added products.
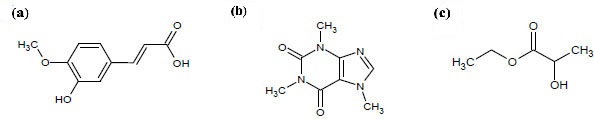
Chemical structure of (a) ferulic acid, (b) caffeine and (c) ethyl lactate.
Ferulic acid (4-hydroxy-3-methoxycinnamic acid) is a phenolic compound found in plant tissues in both free and conjugated form. It is also present in many foods, peel of fruits and vegetables. In most foods, ferulic acid is linked to different molecules, such as hydroxyl acids, mono-, di- and polysaccharides and even sterols [1-3]. The foodstuffs that contain the highest amount of ferulic acid are grains, specially refined corn bran (26-33 mg g-1) [3], barley extract (14-23 mg g-1) [4] and wheat bran (≈ 15 mg g-1) [5]. It has been reported that ferulic acid possess several biological activities. Among them, the antioxidant activity has been the most documented [6, 7].
In addition, ferulic acid could possess pharmacological properties, such as anticancer, anti-atherosclerotic, anti-diabetes and antihypertensive effects [8].
Caffeine (1,3,7-trimethylxanthine) is the most consumed alkaloid in the world. Caffeine is naturally contained in different foods, such as kola nut, guarana, yerba mate and cocoa, but the two most consumed foods rich in caffeine are coffee (9-25 mg g-1) [9] and tea (20-50 mg g-1) [10]. The effects of caffeine as a stimulant of the central nervous system and its adverse health effects are well known. For this reason, there is an increasing demand for decaffeinated products [11]. Decaffeination process using supercritical carbon dioxide is very widespread in the commercial area, although organic solvents are also used. At present, ethyl acetate is preferred due to its lower toxicity [12, 13], as other low toxicity solvents, such as ethanol, ethyl acetate and acetone [14], exhibit poor selectivity when compared to chlorinated solvents [15].
Knowledge of the solubility behavior of ferulic acid and caffeine in water and organic solvents is essential in selecting the most appropriate solvent and in the development of efficient processes for the extraction of these compounds from different vegetable sources.
It has been demonstrated that ferulic acid’s solubility in water is very low (0.08 mass %) [16]. With the view to increase its solubility in aqueous solutions, it has been suggested either to link the acid with different molecules or to form various structures [17, 18]. In this sense, enzymatic catalysis produces lower yields than chemical approach, but the latter uses and produces toxic compounds.
Therefore, the use of selective and efficient agrochemical solvents (either by themselves or mixed with water) for the extraction of such compounds is of great importance.
Ethyl lactate is an agrochemical GRAS (Generally Recognized as Safe) solvent and due to its low toxicity it has been approved by FDA (Food and Drug Administration) and EFSA (European Food Safety Authority) as a pharmaceutical ingredient and food additive. It is used as solvent in different industrial areas owing to its attractive properties, e.g. it is non-corrosive, non-carcinogenic, non-teratogenic, biodegradable and non-ozone depleting. In spite of this, ethyl lactate has been poorly studied as an extraction solvent for food compounds. The research till present has been mainly focused on the extraction and fractionation of lipid related compounds [19], the extraction of fatty acids and carotenoids [20, 21], and on the recovery of caffeine from plant materials [22]. In this case, the data available in the literature regarding the solubility of high-value food compounds is scarce [23].
Manic et al. [24] were the first to examine the influence of water present in small amounts in ethyl lactate on the solubility of several bioactive substances at ambient pressure and in the temperature range (298.2 to 343.2) K and reported a slight increase of caffeine and ferulic acid solubility. In a recent contribution Villanueva-Bermejo et al. [25] reported the solubility of caffeine in ethyl lactate + water mixture at 298 K and atmospheric pressure as a function of the ethyl lactate + water mixture composition, covering the whole range from 100 % ethyl lactate to 100 % water.
In this work, the solubility of ferulic acid in ethyl lactate + water mixtures at the above temperature and pressure conditions, considering the whole range of ethyl lactate + water mixture composition, is measured and reported for the first time The solubility behavior observed is similar to that of caffeine reported previously [25]. Additionally, the experimental data for both systems were correlated applying the activity coefficient models UNIQUAC [26] while the modified (Dortmund) UNIFAC model was applied in a predictive manner [27]. The capabilities of these models to represent correctly the cosolvent effect observed were analyzed and compared.
2. EXPERIMENTAL
2.1. Materials
Trans-ferulic acid (99.0 % purity), ethyl lactate (≥ 98% purity) and acetic acid (≥ 99.0 % purity) were obtained from Sigma–Aldrich (St. Louis, MO, USA). Acetonitrile (HPLC grade) was obtained from Lab-Scan analytical sciences (Gliwice, Poland). Water was high-purity deionized Milli-Q water from Millipore Corporation (Jaffrey, NH, USA).
2.2. Solubility Measurements
The solubility of ferulic acid in the ethyl lactate + water mixtures was measured at 298 K and atmospheric pressure as a function of the ethyl lactate : water ratio (from 0:100 to 100:0 mass %). For all the solutions studied, the liquid mixture and solute in excess were placed into glass vessels (10 ml) with a magnetic stir bar. The vessels were put inside a water bath heated by a magnetic hotplate stirrer (RCT classic IKAMAG® safety control. IKA Works GmbH & Co. KG, Staufen, Germany) which was used to agitate the samples during 24 h under fixed temperature, controlled by an electronic contact thermometer with probe (VWR VT-5 VWR International, LLC West Chester, Pensilvania, USA) with an accuracy of 0.2 K. After reaching the equilibrium, stirring was stopped and the vessels were left still for more than 48 hours to allow a complete phase separation. Stirring and repose times were set as described in our previous work [25] to ensure achieving equilibrium and phase separation. Samples of clear saturated liquid solution were taken and filtered through a 0.20 μm filters and placed into glass vials. Then, samples were appropriately diluted for HPLC analysis.
2.3. HPLC Analysis
The samples were analyzed in a Varian ProStar Analytical HPLC (Agilent Technologies, Santa Clara, CA, USA) with a ternary pump, thermostatic controlled column oven, autosampler mode 410 and diode array detector. All the modules were controlled by PC with interface and HPLC Varian Star system control software.
The injection volume was 10 µl and the column employed was Microsorb-MV100 column C-18, 5 µm (250 mm × 4.6 mm), fitted with a suitable pre-column. The mobile phase used was (A) acetonitrile/(B) 2 % volume acetic acid in water with a flow rate of 0.8 mL/min and 40 ºC of temperature. The gradient employed was as follows: initial 0% A, 10 min 15 % A, 25 min 50 % A, 35 min 100 % A and 40 min 0% A.
The amount of ferulic acid was calculated from a calibration curve using standards and the results were registered at 320 nm.
3. THERMODYNAMIC FRAMEWORK
The equilibrium condition for a pure solid solute (i) dissolved in a liquid phase is:
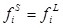 |
(1) |
where fiS is the fugacity of the solute in the solid phase and fiL is the fugacity of the solute in the liquid phase.
An adequate assumption is that the solid phase does not absorb the liquid. Furthermore, provided the solid phase is a pure compound then fiS is the fugacity of the pure solid.
The solute fugacity in the liquid phase can be referred to the fugacity of the pure solute in liquid state fio:
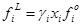 |
(2) |
where γi is the activity coefficient of the solute in the liquid phase and xi is its molar fraction (solubility). fio is typically taken as the pure-liquid fugacity at the system temperature and pure-liquid vapor pressure, with the corresponding corrections for pure-fluid vapor-phase non-ideality, and for the effect of pressure.
Replacing equation (2) in equation (1) the following relation for the solubility is obtained:
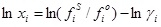 |
(3) |
The ratio fiS / fio can be calculated as follows:
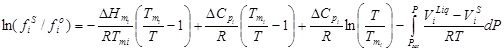 |
(4) |
where Tmi and ΔHmi are, respectively, the solute normal melting temperature and enthalpy of fusion, and ΔCpi is the difference between the heat capacity of the pure liquid and the solid solute.
The first term on the right-hand side of equation (4) is the dominant, and the next two terms tend to cancel each other, especially if the mixture temperature and the solute melting temperature are not far apart. Additionally, the last term of equation (4), which takes into account the effect of pressure on the solute fugacity, is significant only at very high pressures since the difference between the solute molar volume in the liquid and solid states is negligible [28].
Then:
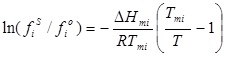 |
(5) |
Taking into account equation (5) and equation (3), the solute solubility can be calculated as follows:
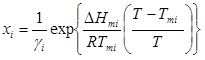 |
(6) |
The above equations set the thermodynamic modeling framework for correlating solid solubilities in liquid mixtures and show that the solute solubility is a function of the solid solute pure compound fusion properties, the system temperature, and the activity coefficient of the solid solute, which is calculated by a thermodynamic model.
In our study the activity coefficients of ferulic acid and caffeine are calculated applying UNIQUAC [26] and the modified UNIFAC models [27].
The normal melting temperature and enthalpy of fusion of ferulic acid were measured experimentally and reported in [29]. The fusion properties of caffeine were taken from [30]. The respective values are given in Table 1.
4. RESULTS AND DISCUSSION
4.1. Experimental Results
The solubility of ferulic acid in ethyl lactate + water mixtures is reported in Table 2. First column of the table indicates the content of water in the binary ethyl lactate + water solvent. Table 2 gives also the Standard Deviation (SD) corresponding to each experimental data point (SD < 0.15). The mean relative SD is 2.8 %.
Solubility of ferulic acid in ethyl lactate + water mixtures. SD: standard deviation.
| Water content (mass %)a | Solubility (mass fraction %) | SD |
|---|---|---|
| 0.00 | 5.34 | 0.08 |
| 9.58 | 9.22 | 0.15 |
| 19.22 | 10.22 | 0.14 |
| 29.22 | 9.52 | 0.11 |
| 38.83 | 7.81 | 0.10 |
| 49.27 | 5.86 | 0.09 |
| 58.93 | 3.78 | 0.11 |
| 69.30 | 1.92 | 0.08 |
| 79.27 | 0.67 | 0.03 |
| 89.46 | 0.19 | 0.01 |
| 100.0 | 0.06 | <0.01 |
Fig. (2) and Fig. (3) show, respectively, the corresponding solubility data for ferulic acid (this work) and caffeine [25]. In both cases, the solubility in the mixed solvent is higher than that in pure ethyl lactate or water. The solubility of ferulic acid and caffeine in water is, respectively, 0.06 and 2.0 mass %. Correspondingly, these solubility values are in a very good agreement with values previously reported in the literature, being 0.08 mass % [16] and 2.2 mass % [15] the solubility reported for ferulic acid and caffeine, respectively. The solubility of ferulic acid and caffeine in pure ethyl lactate is, respectively, 5.3 and 3.0 mass %. Regarding these data, slightly lower values (4.5 and 2.3 mass %, respectively) were obtained by Manic et al. [24] using dried ethyl lactate (<0.03 % mass water), which might be due to the fact that ethyl lactate was not dried before the experiments realized in our study.
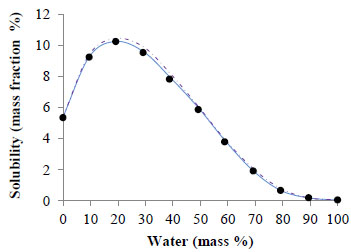
Solubility (mass %) of ethyl lactate + water mixtures: (•) experimental data, ( — ) UNIQUAC and ( -- ) modified UNIFAC correlations.
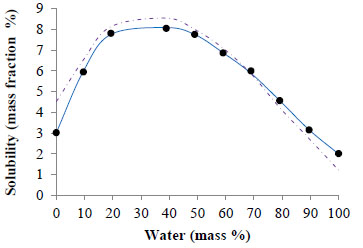
Solubility (mass %) of ethyl lactate + water mixtures: (•) experimental data, ( — ) UNIQUAC and ( -- ) modified UNIFAC correlations.
In the case of ferulic acid, Fig. (2), the solubility attains a maximum of 10.2 % mass for an ethyl lactate + water mixture comprising 20 % of water and is around 1.9 fold higher than the solubility in pure ethyl lactate and 170 fold higher than in water.
The solubility of caffeine, see Fig. (3), attains a maximum of 8.0 % mass for a mixed solvent comprising 40 % mass of water. This solubility value is around 2.7 fold higher than the solubility in pure ethyl lactate and 4.0 fold higher than that in water [25]. The addition of water to the ethyl lactate solvent can substantially increase the solubility of both compounds, supporting the co-solvent effect previously observed by Manic et al. [24].
4.2. Thermodynamic Modeling Results
The activity coefficients of ferulic acid and caffeine in the ethyl lactate + water mixtures were calculated applying UNIQUAC and modified UNIFAC, according to the following general expression:
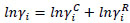 |
(7) |
where γiC and γiR are, respectively, the combinatorial and residual contributions to the activity coefficient γi.
The γiC contribution is calculated exclusively from the pure component chemical parameters, using the relative Van der Waals volumes ri and surface areas qi of the pure chemicals. The γiR contribution accounts to the interactions between molecules present in the system, by means of binary interaction energy parameters (Δuij=uij-uii). In the case of UNIFAC model, these parameters (ri, qi and Δuij) are calculated applying the “solution of groups” concept [27].
The values for the UNIQUAC structural parameters for ferulic acid, caffeine and ethyl lactate used in our study are those reported in ref. [24], while for water are those given in the original reference [26].
The application of UNIQUAC is straightforward provided the binary interaction parameters τij≠τji between the species i and j in the mixture are known. In this work, the binary interaction parameters were considered as adjustable parameters and the goal was to determine values that provided the best fit to the experimental solubility data for both solid solutes. This determination is usually based on some type of least squares or maximum likelihood criterion and requires the solution of a nonlinear optimization problem. It should be pointed out, however, that it is not uncommon for the objective function in such nonlinear parameter estimation problems to be nonconvex and thus to have several local minima. Furthermore, the methods typically used to solve such problems, like the one used in the present study, are local methods that provide no guarantee that the global optimum has been found. Thus, the sets of the binary interaction parameters obtained probably represent just one of the possible sets of parameters. The values for the binary parameters obtained for the two systems of interest are given in Table 3.
The agreement between the experimental solubility data and the UNIQUAC correlation is represented in Fig. (2) (solubility of ferulic acid in ethyl lactate + water mixtures) and Fig. (3) (solubility of caffeine in ethyl lactate + water mixtures).
The Average Absolute Relative Deviation (AARD):
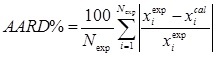 |
(8) |
is lower than 1.5 %, which demonstrates the excellent quantitative performance of the UNIQUAC model in both cases examined.
The application of the modified UNIFAC model is fully predictive and is unproblematic for the case of ferulic acid solubility in the mixed solvents, as all required functional groups and the respective group-group interaction parameters for ferulic acid, ethyl lactate and water are available in the parameter matrix (see Table 4). The representation of caffeine, however, is far from straightforward due to the lack of parameters for the cycl-CO group. Thus, caffeine molecule was instead represented as a combination of existing functional groups that we considered most closely resembles it. This representation, of course, is not precise. Moreover, as there are other possible representations it should be underlined that different choices of UNIFAC functional groups will yield different predictions. The latter is just one more illustration of a major uncertainty associated with the use of UNIFAC.
Functional groups chosen to represent the chemical structure of the solutes, caffeine and ferulic acid, and the solvents, ethyl lactate and water.
| Caffeine | Ferulic acid | Ethyl lactate | Water | ||||
|---|---|---|---|---|---|---|---|
| Group | Number of groups | Group | Number of groups | Group | Number of groups | Group | Number of groups |
| c-C=C | 1 | CH=CH | 1 | CH3 | 2 | H2O | 1 |
| c-CON-CH3 | 2 | ACH | 3 | CH2 | 1 | ||
| CH3N | 1 | AC | 2 | CH | 1 | ||
| ACNH | 1 | ACOH | 1 | OH (s) | 1 | ||
| COOH | 1 | COO | 1 | ||||
| CH3O | 1 | ||||||
The agreement between the experimentally measured and modified UNIFAC (Dortmund) predicted solubility of ferulic acid in ethyl lactate + water mixtures is very good with an AARD of 4.8 % (see Fig. 2). For caffeine (Fig. 3), however, the agreement is not very good, with an AARD of 14.0 % and hence should be considered rather qualitative than quantitative.
CONCLUSION
In this work the solubility of ferulic acid in ethyl lactate + water mixtures, at ambient temperature and pressure, was measured as a function of the mixed solvent composition and reported for the first time. The solubility pattern exhibited is similar to that reported for caffeine in a previous work [25] with a distinctive maximum. The cosolvent effect observed maybe of interest from an economical point of view in order to enlarge the extraction of these bioactives from vegetal raw materials.
The solubility data for ferulic acid and caffeine were represented applying UNIQUAC and modified UNIFAC models. UNIQUAC demonstrated excellent correlative capacity for both systems studied with an overall AARD < 1.5 %). UNIFAC (Dortmund) was applied in a completely predictive manner; the results obtained for ferulic acid are very good while for caffeine should be considered just qualitative.
CONFLICT OF INTEREST
The authors confirm that this article content has no conflict of interest.
ACKNOWLEDGEMENTS
D. Villanueva acknowledges the predoctoral contract (JAE) given by the Consejo Superior de Investigaciones Científicas (CSIC) of Spain. This work was financed thanks ALIBIRD, S2013/ABI-2728 (Comunidad de Madrid) project.


